Improved formulation screening and local dermal PK and PD using dOFM sampling
Dermal drug development and bioequivalence studies for generics are limited by uninformative blood PK parameters. PD and PK measurements directly at the site of action provide much more valuable information for formulation selection and efficacy testing. Another essential criterion in successful drug development are reliable data on protein binding prediction in the skin.
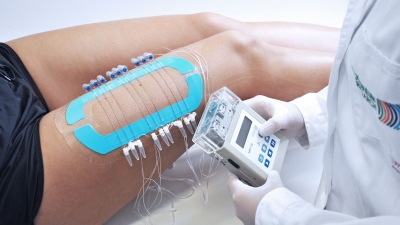
Dermal open flow microperfusion (dOFM) technology allows us to continuously monitor the dermal interstitial fluid. dOFM samples are a direct representation of the sampled dermal tissue and include all substances (even living cells and highly lipophilic compounds) in a diluted form. dOFM is also capable of introducing substances into the skin thus enabling microdosing approaches for solid clinical data in very early drug development stages.
Profit from temporally- and spatially-resolved dermal PK monitoring for direct comparison of formulations in one subject. This provides reliable data from early preclinical drug development to clinical bioequivalence testing of dermal topical products.
This webinar will give you a concise overview of dOFM capabilities and possible setups for preclinical and clinical studies. We will present several case studies for drug development, biomarker discovery and bioequivalence assessment. Our experts will personally be present to answer all your questions.
To View & Download the Presentation Slides & Speaker Notes, Please Click Here
To View & Download the Full Size Presentation Slides, Please Click Here
To View & Download the Q&A Catalogue, Please Click Here
Presented by

Dr. Thomas Birngruber,
Head of Research Group Biomedical Tissue Monitoring
Dr. Birngruber has a background in biomedical engineering, inter-disciplinary medical research and in-vivo sampling techniques. He has more than 15 years of experience with the use of Open Flow Microperfusion OFM and other sampling techniques in different tissues (brain, dermal and adipose tissue) and has led the optimization and standardization of dermal OFM in the last few years.

Dr. Frank Sinner,
Director
Dr. Sinner has a background in analytical chemistry and has addressed challenges of in-vivo sampling for the last 20 years. As director of HEALTH – Institute for Biomedicine and Health Sciences he led the certification of dOFM for human use and has advanced dOFM as a method for bioequivalence studies for topical, dermal generic drugs.






